 
They readily gain one electron to achieve a stable octet configuration. For elements after No, the electron configurations are tentative. Elements in Group 17, known as halogens, have a valency of -1. Figure 1: Periodic Table (Free Printable periodic tables (pdf and PNG) 2023) Explain the significance of the S, P and D blocks in the periodic table. The periodic table shows the atomic mass and atomic number of each element. According to Coulombs law, the attraction of an electron to a nucleus depends only on three factors: the charge of the nucleus (+Z), the charge of the electron (-1), and the distance between the two ((r)). Take a closer look at the some of the elements in the periodic table’s far right column in Figure 3. The ideal gas law is easy to remember and apply in solving problems, as long as you get the proper values a. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located. (b) Covalent radii of the elements are shown to scale. This rule works only for elements excluding the transition metals. The paper introduces Chemdex, a freely accessible web-based database of over 70 000 compounds characterised by crystallography from across the periodic table. 
To find how many valence electrons are in an element, simply locate the column number that it is in, and that determines the number of valence electrons in an element. The atomic radius for the halogens increases down the group as n increases. The valence, in Mendeleevs understanding, was an indication of the ability of an element to combine with other elements. The periodic table contains rows and columns. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. 
In the second period elements, the two electrons in the 1s 1 s sublevel are called inner-shell electrons and are not involved directly in the elements reactivity, or in the formation of compounds. 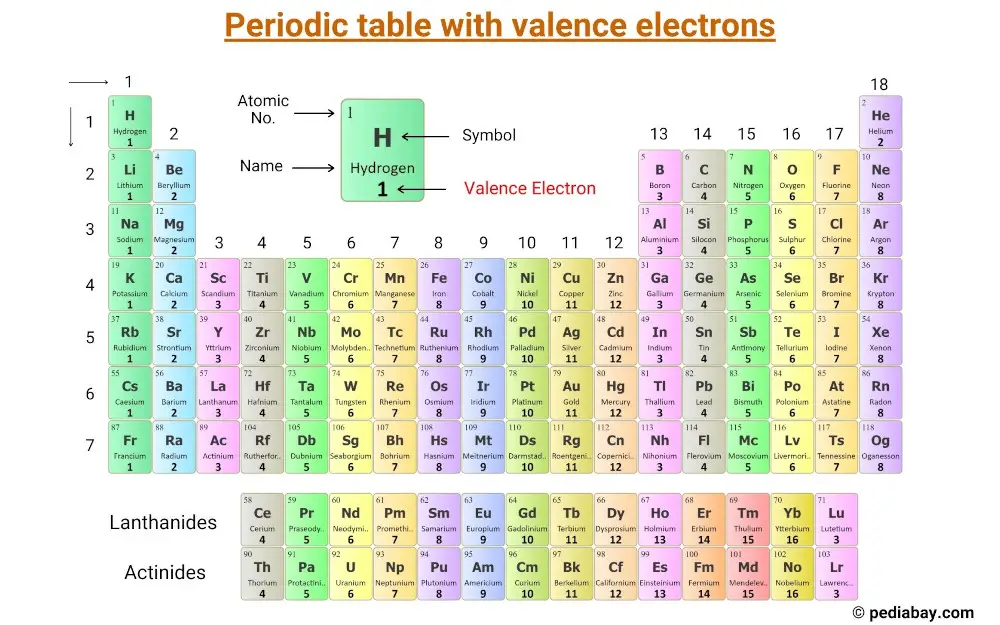
For hydrogen, there is only one electron and so the nuclear charge ( Z) and the effective nuclear charge ( Z eff) are equal.\): Electron Configurations of the Elements. Valence electrons are the electrons in the highest occupied principal energy level of an atom. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. For the transition element, the valence electron has to be determined by adding the total electrons of the d-orbital to the electron in the last shell of the atom. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
